BY CHILLYMANJARO
Ajinomoto, maker of aspartame, turns to new malicious strategy which hopes to trick public with renaming and marketing it as natural. Artificial sweetener Aspartame is now rebranded and its called “AminoSweet“.
Over 25 years ago, aspartame was first introduced into the European food supply. Today, it is an everyday component of most diet beverages, sugar-free desserts, and chewing gums in countries worldwide. But the general public is waking up to the truth about artificial sweeteners like aspartame and the harm they cause to health. The latest aspartame marketing scheme is a desperate effort to indoctrinate the public into accepting the chemical sweetener as natural and safe, despite evidence to the contrary.
Despite the myriad of evidence gained over the years showing that aspartame is a dangerous toxin, it has remained on the global market with the exception of a few countries that have banned it. In fact, it continued to gain approval for use in new types of food despite evidence showing that it causes neurological brain damage, cancerous tumors, and endocrine disruption, among other things.
History of aspartame
The details of aspartame’s history are lengthy, but the point remains that the carcinogen was illegitimately approved as a food additive through heavy-handed prodding by a powerful corporation with its own interests in mind. Practically all drugs and food additives are approved by the FDA not because science shows they are safe but because companies essentially lobby the FDA with monetary payoffs and complete the agency’s multi-million dollar approval process.
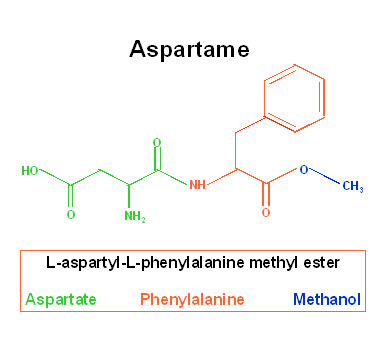
Aspartame was an accidental discovery by James Schlatter, a chemist who had been trying to produce an anti-ulcer pharmaceutical drug for G.D. Searle & Company back in 1965. Upon mixing aspartic acid and phenylalanine, two naturally-occurring amino acids, he discovered that the new compound had a sweet taste. The company merely changed its FDA approval application from drug to food additive and aspartame was born.
G.D. Searle & Company first patented aspartame in 1970. An internal memo released in the same year urged company executives to work on getting the FDA into the “habit of saying yes” and of encouraging a “subconscious spirit of participation” in getting the chemical approved.
G.D. Searle & Company submitted its first petition to the FDA in 1973 and fought for years to gain FDA approval, submitting its own safety studies that many believed were inadequate and deceptive. Despite numerous objections, including one from its own scientists, the company was able to convince the FDA to approve aspartame for commercial use in a few products in 1974, igniting a blaze of controversy.
In 1976, then FDA Commissioner Alexander Schmidt wrote a letter to Sen. Ted Kennedy expressing concern over the “questionable integrity of the basic safety data submitted for aspartame safety”. FDA Chief Counsel Richard Merrill believed that a grand jury should investigate G.D. Searle & Company for lying about the safety of aspartame in its reports and for concealing evidence proving the chemical is unsafe for consumption.
How Aspartame Became Legal – The Timeline
Changing aspartame’s name to something that is “appealing and memorable”, in Ajinomoto’s own words, may hoodwink some but hopefully most will reject this clever marketing tactic as nothing more than a desperate attempt to preserve the company’s multi-billion dollar cash cow. Do not be deceived.(NaturalNews)
Artificial sweeteners are linked to behavioral problems, hyperactivity, and allergies. Because saccharin was shown to increase the incidence of bladder cancer in animal testing, all foods containing this sugar substitute are required to carry a warning label. (Puristat)
Link betweenAspartame and Gulf War Syndrome
When stored or heated above 30 degrees Celsius (85 degrees Fahrenheit), as in desert heat, Aspartame in Diet Drinks breaks down into neurotoxic substances: Methanol (wood alcohol), Formaldehyde (embalming fluid), Formic Acid (ant sting venom) and DKP(brain tumor agent).
The major soft drink companies shipped free Diet Drinks to US Desert Storm troops, where they broke down in storage into highly toxic substances. The symptoms reported by the sufferers of “Gulf War Syndrome” mimic the ones of those reporting problems associated with Aspartame use here … but, the military personnel symptoms are greater because they were drinking/eating “expired Diet Pepsi and then some!” Since Aspartame is capable of changing the DNA (in lab tests), birth defects in offspring can be readily addressed. Family members are also experiencing illness, possibly from their continued use of these products. What is the real reason behind Gulf War Syndrome? Based on volumes of available research, we believe it was what they were eating and drinking in extreme desert heat.
ACSN Founder, Mary Nash Stoddard (a veteran’s widow) is available to answer questions and support allegations regarding this important issue and how it is adversely affecting 3 out of 5 who use it. ACSN’s special Pilot’s Hotline has logged over 800 pilot-related calls. Pilots are having grand mal seizures in the cockpits of commercial airline flights, and others are crashing the flight simulator at training facilities while in seizure. Dozens have lost their flying status because of Aspartame. Seizures, loss of vision, vertigo, suicidal depressions, heart problems, etc. are routinely reported to ACSN. Many consumers report bizarre psychological symptoms as well. This happens when the Phenylalanine in Aspartame depletes levels of the neurotransmitter, Serotonin, a brain chemical which regulates aggressive behaviors and sleep patterns, among other things.
The U.S. Air Force has formally warned all pilots to abstain from consuming Diet Drinks in their official Flying Safety publication (May & August 1992.) Their Aspartame Alert article was based on that of the U.S. Navy to pilots earlier in Navy Physiology. 78% of all consumer complaints to the FDA are about reactions to Aspartame (NutraSweet/Equal.)
Recently, a letter was sent to COPA in which a pilot expressed his concerns over questionable adverse effects from consumption of diet drinks containing aspartame. By 1986, the FDA and the Centre for Disease Control in the U.S. had evaluated 3,000 known complaints.
The article disclosed that methanol destroys the brain, albeit a little at a time, and that effects are cumulative. Depending on a person’s physical state and tolerance level, immediate effects can either be severe (resulting in epileptic seizures, including grand mal, blindness, chest palpitations), or less noticeable (causing blurred vision, bright flashes, tunnel vision, ringing or buzzing in ears, migraine headaches, dizziness, loss of equilibrium, lip and mourth reactions); less noticeable effects might be passed off as temporary or caused by something else. But everyone is affected in one way or another, since methanol causes toxic reactions, not just allergic ones in a few unfortunates.
If pilots want more information, call (or Email) the Aspartame Consumer Safety Network in Dallas, Texas U.S.A. and talk to ACSN & Worldwide Pilot Hotline Founder, Mary Nash Stoddard [1-214-352-4268 or Email: marystod@airmail.net] ACSN promises confidentiality if asked, and will send an eye-opening information package.
Apparently, the other main components of Aspartame, Phenylalanine [50%], Aspartic Acid [40%], combined with the Methanol [10%], create a witch’s brew of 16 breakdown products after digestion that cause illness. Animal tests revealed brain tumors, some cancerous; holes in the brain, womb tumors, uterine tumors and reproductive dysfunctions. Studies on humans indicated that pregnant women and young children run especially high risks. There is evidence of birth defects in offsprings, including severe retardation, genetic damage, especially in women who carry the PKU (Phenylketonuria) gene, heart problems and emotional and psychiatric disorders.
Aspartame (Nutrasweet) Toxicity Information Center
Aspartame Dangers and Side Effects
DORway » Aspartame & Aspartame Poisoning Information Site
Adverse reactions and side effects of aspartame include:
Eye
blindness in one or both eyes
decreased vision and/or other eye problems such as: blurring, bright flashes, squiggly lines, tunnel vision, decreased night vision
pain in one or both eyes
decreased tears
trouble with contact lenses
bulging eyes
Ear
tinnitus – ringing or buzzing sound
severe intolerance of noise
marked hearing impairment
Neurologic
epileptic seizures
headaches, migraines and (some severe)
dizziness, unsteadiness, both
confusion, memory loss, both
severe drowsiness and sleepiness
paresthesia or numbness of the limbs
severe slurring of speech
severe hyperactivity and restless legs
atypical facial pain
severe tremors
Psychological/Psychiatric
severe depression
irritability
aggression
anxiety
personality changes
insomnia
phobias
Chest
palpitations, tachycardia
shortness of breath
recent high blood pressure
Gastrointestinal
nausea
diarrhea, sometimes with blood in stools
abdominal pain
pain when swallowing
Skin and Allergies
itching without a rash
lip and mouth reactions
hives
aggravated respiratory allergies such as asthma
Endocrine and Metabolic
loss of control of diabetes
menstrual changes
marked thinning or loss of hair
marked weight loss
gradual weight gain
aggravated low blood sugar (hypoglycemia)
severe PMS
Other
frequency of voiding and burning during urination
excessive thirst, fluid retention, leg swelling, and bloating
increased susceptibility to infection
Additional Symptoms of Aspartame Toxicity include the most critical symptoms of all
death
irreversible brain damage
birth defects, including mental retardation
peptic ulcers
aspartame addiction and increased craving for sweets
hyperactivity in children
severe depression
aggressive behavior
suicidal tendencies
Aspartame may trigger, mimic, or cause the following illnesses:
Chronic Fatigue Syndrome
Epstein-Barr
Post-Polio Syndrome
Lyme Disease
Grave’s Disease
Meniere’s Disease
Alzheimer’s Disease
ALS
Epilepsy
Multiple Sclerosis (MS)
EMS
Hypothyroidism
Mercury sensitivity from Amalgam fillings
Fibromyalgia
Lupus
non-Hodgkins
Lymphoma
Attention Deficit Disorder (ADD)